- Messages
- 438
- Reaction score
- 3,645
- Points
- 253
We are currently struggling to cover the operational costs of Xtremepapers, as a result we might have to shut this website down. Please donate if we have helped you and help make a difference in other students' lives!
Click here to Donate Now (View Announcement)
Kindly post them here as soon as you get them.m looking for em too
but they wont officially be released b4 the 17th :/
can u pls explain the charges on each?
It can form only coordination 4 which shows cis trans isomer . For cis chlorine should be 90 degree to each other while for trans both chlorine should be 180 degree to each other . Chorine placed anywhere other then 180 degree will only have 90 degree because all four are on same plane. Not to loose marks just male chlorine next to the other chlorine to show cis isomer.View attachment 63545
How do we come to know where to place the Cl and NH3? Cant there be any other arrangement apart from these?
View attachment 63546
Im sure you calculated the Mr for i.
Charges? I didn't get itcan u pls explain the charges on each?
its simply the number of proton attached to the carbon if its CH3 the relative peak area is 3 if OH the 1 and so on
Simply!!!its simply the number of proton attached to the carbon if its CH3 the relative peak area is 3 if OH the 1 and so on
Also Holmes this is actually the correct explanationAnyone?
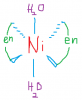
Zaki ali asgharcan u pls explain the charges on each?
I dont think this is the case. It would still show optical isomerism.if there were 3 en ligands you could have done optical but in this case its 2 different ligands 2 en's and one other ligand so its cis trans
Great shotAlso Holmes this is actually the correct explanation
well you can only have two isomers
cis and trans
and for the cis isomer, there will be two optical isomers since there is also a bidentate ligand present
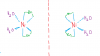
THIS IS TRANS
View attachment 63567
THE ONES BELOW ARE BOTH CIS AND MIRROR IMAGES OF EACH OTHER...THE LINE IN BETWEEN IS THE MIRROR
THIS KIND OF ISOMERISM IS ONLY POSSIBLE WHEN BIDENTATE LIGANDS ARE ALSO PRESENT
View attachment 63568
just like HCOOH is further oxidised to CO2 by KMnO4, similarly, HOOCCOOH is also oxidisedWhy would HO2CCO2H react with MN04- ions? and which reaction is this?
well the first one shows both: optical due to bidentates and cis trans because there are pairs of two types of ligands.This one too.
For almost 10 years, the site XtremePapers has been trying very hard to serve its users.
However, we are now struggling to cover its operational costs due to unforeseen circumstances. If we helped you in any way, kindly contribute and be the part of this effort. No act of kindness, no matter how small, is ever wasted.
Click here to Donate Now