- Messages
- 59
- Reaction score
- 130
- Points
- 43
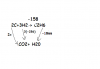
you did something wrong , the equation of reaction is PbCl4 +2NaBr--> Pbcl2+Br2+ 2NaCl.ok,
according to the data,
the equation of the reaction is.
PbCl4 +4NaBr-->Pb+2Br2+4NaCl.
now
6.980g of PbCl4
calculate its moles,Mr=349
6.980/349
=0.02mol
now since mol ration is 1:2,
we have 0.04mol of Bromine.
to get the mass.
multiply moles with its mr
= 0.04X79.9
=3.196
^ your answer.
moles of PbCl4 is 6.98/349= 0.02
1:1 ratio = 0.02 mole of Br2
multiply 0.02 by mr of Br2 = 0.02X(79.9+79.9)= 3.196