- Messages
- 12
- Reaction score
- 43
- Points
- 23
Can you please help me with these as well


Answers are B and D


Answers are B and D
We are currently struggling to cover the operational costs of Xtremepapers, as a result we might have to shut this website down. Please donate if we have helped you and help make a difference in other students' lives!
Click here to Donate Now (View Announcement)


thank you I thought it was the same atom that was broken..I2 (solid) --> I2 (gas)
The bond broken is from one I2 molecule to other I2 molecules, which is induced dipole (as I2 are non polar).
Covalent bonds are NOT broken as the molecules are not broken into atoms.
which exam is this?
Thank you so so much!!!
You didn't actually get my point though I needed explanation only on 11, 32, 33 and 35.
But anyway thanks so much for taking your time and explaining everything it might be of good use in the end.
Q1. Find out the number of moles of O2, since 1 mole of gas occupies 24000 cm^3 at RTP.
moles of O2 = 500/24 000
molecules of O2 = moles x 6.02 x 10^23 = (500/24000) x 6.02 x 10^23 = 1.25 x 10^22
Q2. Comparing the original structure to the resulting structure, we see that 5 C=C double bonds "gone".
So 5 moles of H2 are added.
If unable to visualise the above explanation, the diagram can show how H could be added in these locations.
View attachment 44133
Q7 and Q10
View attachment 44134
Q21. Is the answer really D? I got B.
View attachment 44136
Q22. The C=C undergoes oxidative cleaving, and the ends are oxidised to COOH.
View attachment 44137
Q23.
If confident, this question can be approached in a mathematical way
CnH(2n+2) + (3n+1)/2 O2 --> n CO2 + (n+1)H2O
when n increases, moles of O2 increases linearly. So its a line with a positive gradient.
Can you please help me with these as well

Answers are B and D
http://papers.xtremepapers.com/CIE/Cambridge International A and AS Level/Chemistry (9701)/9701_s04_qp_1.pdf
Q9
Q35 isn't 3 suppose to be correct too?
Q37
Please anyone Q39. I cant understand a word.
http://papers.xtremepapers.com/CIE/Cambridge International A and AS Level/Chemistry (9701)/9701_w08_qp_1.pdf
B is correct.
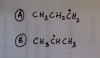
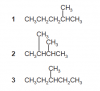
Can someone please help me in this question
May 2012 Paper 12 Q23
I would have definitely attached the link but I am from my mobile
Thanks in advance!!!
Thanks!!number of moles of Tl+NO3-=10*0.30/1000
=3*10^-3
Number of moles of NH4+VO3-=20*0.10/1000
=2*10-3
Divide by the smalles figure, we would obtain the ratio of 1.5:1 *2 to get rid of a fraction so the mole ration will 3:2 so for every 3 moles of Tl+( six electrons are removed) only two moles are reduced so divide the six electrons by 2 to figure out how many electrons are gained per mole
Q15. This is a similar to question Q14 of 2006.
Group II nitrates decompose based on the equation below:
2 X(NO3)2 (s) --> 2 XO (s) + 4 NO2 (g) +O2(g)
1.32 g is mass of NO2 and O2 gas, the left over mass 0.68 g is XO.
Mass of XO = 0.68g
Moles of XO = 0.68/ (Mr of X + 16)
Mass of X(NO3)2 = 2 g
Moles of X(NO3)2 = 2/(Mr of X + 62)
since moles of X(NO3)2 = moles of XO
2/(Mr of X + 124) = 0.68/ (Mr of X + 16)
We can solve for Mr of X using the normal maths approach, but it might be faster to do trial and error from the Mr of the four options.
Be : 2/(9 + 124) does not equal to 0.68/ (9 + 16)
Ca: 2/(40 + 124) = 0.68/ (40+ 16)
Therefore, answer is Ca.
Q17. At RTP, 1 mol of gas (assume ideal) occupies 24 000 cm3.
mole of O2 = 300 /24 000 = 0.0125 mol
2Ca + O2 --> 2 CaO
2Mg + O2 --> 2 MgO
4K + O2 --> 2K2O
4Na + O2 --> 2 Na2O
0.0125 mol of O2 will react with:
0.025 mol of Ca (1 g)
0.05 mol of Mg (1.2 g)
0.05 mol of K (1.95 g)
0.05 mol of Na (1.15 g)
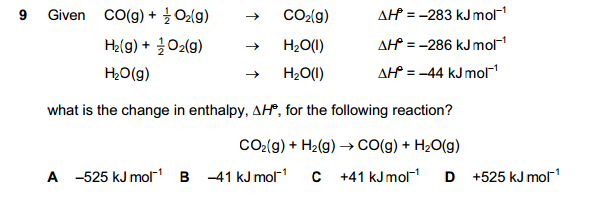
For almost 10 years, the site XtremePapers has been trying very hard to serve its users.
However, we are now struggling to cover its operational costs due to unforeseen circumstances. If we helped you in any way, kindly contribute and be the part of this effort. No act of kindness, no matter how small, is ever wasted.
Click here to Donate Now