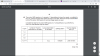
how can we determine element b and c are in group 2 and 1,but not group 12 and 11? should we refer to data booklet for checking the standard 1st ionisation energies to get the elements and correct group?
You're looking for a jump in successive ionization energies, which means that all the outermost electrons were removed and the next one is in an inner shell.
B has a jump between two and three, which means that it has two valence electrons, group 2.
C has a jump between one and two, one valence electron, group 1.