-
We need your support!
We are currently struggling to cover the operational costs of Xtremepapers, as a result we might have to shut this website down. Please donate if we have helped you and help make a difference in other students' lives!
Click here to Donate Now (View Announcement)
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Chemistry: Post your doubts here!
- Thread starter XPFMember
- Start date
- Messages
- 3,937
- Reaction score
- 14,646
- Points
- 523
y r tertiary alcohols resistent to oxidation?
how is the substitution reaction of an alcohol wit a hydrogen halide a nucleophilic substitution?cuz as far as i noe -nucleophiles are electron donors so wer is the donation of electrons taking place here??
how is the substitution reaction of an alcohol wit a hydrogen halide a nucleophilic substitution?cuz as far as i noe -nucleophiles are electron donors so wer is the donation of electrons taking place here??
Ok i dont mean to do spam or anything but please go to the following thread and make dua for all of us :
http://www.xtremepapers.com/community/threads/dua-for-all-candidates.14272/
Dont forget to share and Remember every share makes a difference
http://www.xtremepapers.com/community/threads/dua-for-all-candidates.14272/
Dont forget to share and Remember every share makes a difference
- Messages
- 6,440
- Reaction score
- 31,077
- Points
- 698
Did u try googling it??y r tertiary alcohols resistent to oxidation?
how is the substitution reaction of an alcohol wit a hydrogen halide a nucleophilic substitution?cuz as far as i noe -nucleophiles are electron donors so wer is the donation of electrons taking place here??
nywy...The answer to this question depends on your point of view. From a thermodynamic point of view tertiary alcohols are resistant to oxidation, due to the fact that C-C single bonds are stable. Simply speaking, a carbon atom surrounded by three further carbon atoms and a C-O single bond will be more stable than a carbon atom surrounded by two carbon atoms and a C=O group. Quantum mechanical (ab-initio) calculations show that C-C single bonds are slightly electron donating with respect to each other, this means that the central carbon atom of a tertiary alcohol will be more stable than its oxidised analogue - therefore tertiary alcohols are resistant to oxidation under standard conditions.
One thing to note! Tertiary alcohols are only resistant to oxidation under standard conditions with mild oxidising agents - oxidation of tertiary alochols is possible, albeit under quite extreme conditions.
- Messages
- 375
- Reaction score
- 205
- Points
- 53
Q-6, 8, 9, 10, 18, 25, 35
http://www.xtremepapers.com/papers/... AS Level/Chemistry (9701)/9701_s10_qp_11.pdf
http://www.xtremepapers.com/papers/... AS Level/Chemistry (9701)/9701_s10_ms_11.pdf
i believe its the 3rd time i'm posting :/
http://www.xtremepapers.com/papers/... AS Level/Chemistry (9701)/9701_s10_qp_11.pdf
http://www.xtremepapers.com/papers/... AS Level/Chemistry (9701)/9701_s10_ms_11.pdf
i believe its the 3rd time i'm posting :/
- Messages
- 3,937
- Reaction score
- 14,646
- Points
- 523
thnxxxx....dat helped..Did u try googling it??
nywy...The answer to this question depends on your point of view. From a thermodynamic point of view tertiary alcohols are resistant to oxidation, due to the fact that C-C single bonds are stable. Simply speaking, a carbon atom surrounded by three further carbon atoms and a C-O single bond will be more stable than a carbon atom surrounded by two carbon atoms and a C=O group. Quantum mechanical (ab-initio) calculations show that C-C single bonds are slightly electron donating with respect to each other, this means that the central carbon atom of a tertiary alcohol will be more stable than its oxidised analogue - therefore tertiary alcohols are resistant to oxidation under standard conditions.
One thing to note! Tertiary alcohols are only resistant to oxidation under standard conditions with mild oxidising agents - oxidation of tertiary alochols is possible, albeit under quite extreme conditions.
ps.nope i dint try googling it..!! ;P
- Messages
- 6,440
- Reaction score
- 31,077
- Points
- 698
its a nucleophilic substitution bcoz d C atom bonded to the hydroxyl grp will carry a partially positive charge (diff in electronegativity of O n C).how is the substitution reaction of an alcohol wit a hydrogen halide a nucleophilic substitution?cuz as far as i noe -nucleophiles are electron donors so wer is the donation of electrons taking place here??
This makes it open to nucleophilic attack. ie the partially -ve halogen atom in hydrogen halide will attack the bond btw the C n hydroxyl grp..
Hope i helped!!
- Messages
- 3,937
- Reaction score
- 14,646
- Points
- 523
yaaa that helped..!! A LOOT!!its a nucleophilic substitution bcoz d C atom bonded to the hydroxyl grp will carry a partially positive charge (diff in electronegativity of O n C).
This makes it open to nucleophilic attack. ie the partially -ve halogen atom in hydrogen halide will attack the bond btw the C n hydroxyl grp..
Hope i helped!!
(i kinda tend to miss the most obvious stuff..!!
thnx once again
- Messages
- 198
- Reaction score
- 47
- Points
- 28
Ammonium nitrate, NH4NO3, can decompose explosively when heated.
NH4NO3 → N2O + 2H2O
What are the changes in the oxidation numbers of the two nitrogen atoms in NH4NO3 when this
reaction proceeds?
A –2, –4 B +2, +6 C +4, –6 D +4, –4
NH4NO3 → N2O + 2H2O
What are the changes in the oxidation numbers of the two nitrogen atoms in NH4NO3 when this
reaction proceeds?
A –2, –4 B +2, +6 C +4, –6 D +4, –4
- Messages
- 84
- Reaction score
- 49
- Points
- 28
For Question 11 you can see that the equilibrium concentration of Z decreases as the temperature is increased. This means that increasing the temperature lowers the forward reaction (or more simply the Kc for the reaction), and you must remember that Kc for exothermic reaction decreases with increasing temperature (because exothermic gives out heat energy)Anyone explain these two questions to me please? Thanks first! :Yahoo!:
FYI, the answer given for Q10 is D whereas for Q11 is D.


Therefore answer is D.
- Messages
- 6,440
- Reaction score
- 31,077
- Points
- 698
yaaa that helped..!! A LOOT!!
(i kinda tend to miss the most obvious stuff..!!)
thnx once again
- Messages
- 84
- Reaction score
- 49
- Points
- 28
Ammonium nitrate, NH4NO3, can decompose explosively when heated.
NH4NO3 → N2O + 2H2O
What are the changes in the oxidation numbers of the two nitrogen atoms in NH4NO3 when this
reaction proceeds?
A –2, –4 B +2, +6 C +4, –6 D +4, –4
Answer is D.
First check the oxidation of Nitrogen atom in NH4(+1)
x+4=1 so x= -3
Oxidation of Nitrogen in Ammonium Ion = -3
Now Oxidation of Nitrogen in NO3(-1)
x-6=-1 so x= 5
Oxidation of Nitrogen in Nitrate Ion is 5
Now Check the Oxidation State of Nitrogen in N20.
2x-2=0 so x=1
So Oxidation of Nitrogen in N20 is 1
Final Step :
Calculate the change : from -3 to +1 ( change of +4)
from 5 to +1 (change of -4)
Therefore answer is D
- Messages
- 6,440
- Reaction score
- 31,077
- Points
- 698
For 8, find the mass of Cl2 ie 5.287 - 2.920 = 2.367gQ-6, 8, 9, 10, 18, 25, 35
http://www.xtremepapers.com/papers/... AS Level/Chemistry (9701)/9701_s10_qp_11.pdf
http://www.xtremepapers.com/papers/... AS Level/Chemistry (9701)/9701_s10_ms_11.pdf
i believe its the 3rd time i'm posting :/
thn find the no of moles of Cl2 ie ( 2.367/71) o.o33 moles
we know the ratio of moles of X : Cl2 is 1:1 thus no of moles of X is also 0.033
then simply substitute moles n mass in Moles = Mass/ Mr , we get Mr = 87.6 which is the mass no of Strontium...so D is the ans..
- Messages
- 301
- Reaction score
- 114
- Points
- 53
Can anyone explain this question?Q21 How does the compound have 5 double bonds. I counted 6(one in the side chain and 5 in the cyclohexene ring due to presense of just one hydrogen in it.)
Did it for you (sorry, diagram doesn't really look good)
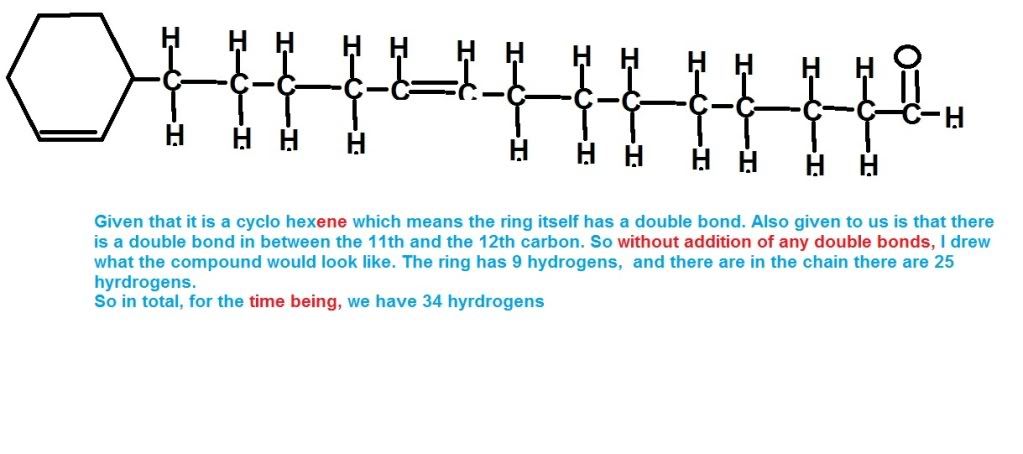
Then
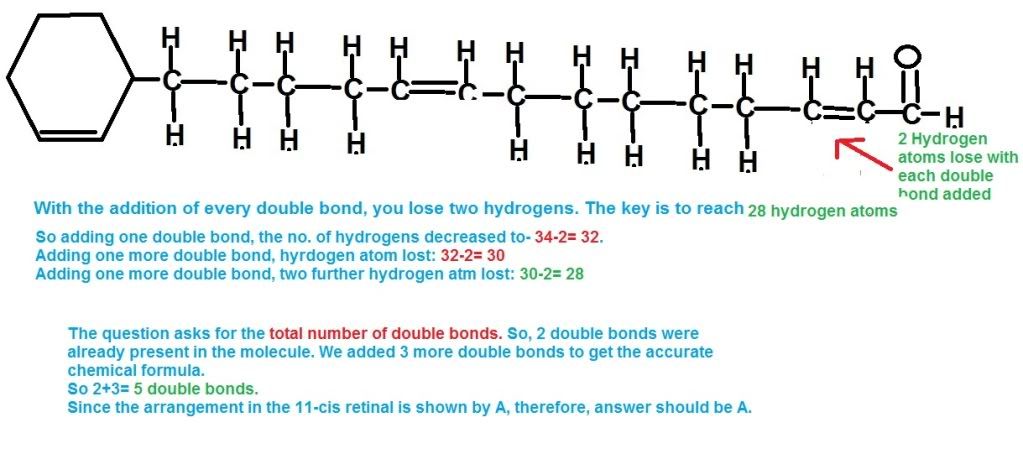
- Messages
- 6,440
- Reaction score
- 31,077
- Points
- 698
For 9, 1 mole of gas = 24 dm3
x moles of gas = 3 dm3....ie 0.125 moles = 3 dm3
Now find the no of moles of all the gases given... Mass/Mr
the ans is C
x moles of gas = 3 dm3....ie 0.125 moles = 3 dm3
Now find the no of moles of all the gases given... Mass/Mr
the ans is C
- Messages
- 6,440
- Reaction score
- 31,077
- Points
- 698
For 10, look at the pH values given for X n Y...
This shows that X is an acid - pH =pH below 7, n Y is a base pH abv 7.
Student P says that X is a strong acid which is completely wrong, as strong acids do not have such a high pH..
However Q says tht Y dissociates more, n since Y is a base, we shud know tht the greater the value of pH, the stronger the acid..
Y is 2 nos greater thn 7, n X is only 1 no lesser thn 7...this shows tht Y is a stronger base n dissociates more..
Hence Q only is correct..
This shows that X is an acid - pH =pH below 7, n Y is a base pH abv 7.
Student P says that X is a strong acid which is completely wrong, as strong acids do not have such a high pH..
However Q says tht Y dissociates more, n since Y is a base, we shud know tht the greater the value of pH, the stronger the acid..
Y is 2 nos greater thn 7, n X is only 1 no lesser thn 7...this shows tht Y is a stronger base n dissociates more..
Hence Q only is correct..
- Messages
- 375
- Reaction score
- 205
- Points
- 53
Ammonium nitrate, NH4NO3, can decompose explosively when heated.
NH4NO3 → N2O + 2H2O
What are the changes in the oxidation numbers of the two nitrogen atoms in NH4NO3 when this
reaction proceeds?
A –2, –4 B +2, +6 C +4, –6 D +4, –4
leosco1995 had answerd this..
Figure out the oxidation numbers in both N atoms in NH4NO3 and compare it with the oxidation number of N in N2O. Like so:
You split up NH4NO3 into the NH4+ and NO3- ions.
In NH4+, the charge on Nitrogen is 1 - 4 = -3.
In NO3-, the charge on Nitrogen is -1 + 6 = +5.
And in N2O, the charge on nitrogen is 2/2 = +1.
And the changes in these are +4 and -4 respectively, meaning D is right.
- Messages
- 3,937
- Reaction score
- 14,646
- Points
- 523
the greater the ph value the weaker the acid..rit?For 10, look at the pH values given for X n Y...
This shows that X is an acid - pH =pH below 7, n Y is a base pH abv 7.
Student P says that X is a strong acid which is completely wrong, as strong acids do not have such a high pH..
However Q says tht Y dissociates more, n since Y is a base, we shud know tht the greater the value of pH, the stronger the acid..
Y is 2 nos greater thn 7, n X is only 1 no lesser thn 7...this shows tht Y is a stronger base n dissociates more..
Hence Q only is correct..
- Messages
- 321
- Reaction score
- 331
- Points
- 73
Ya noww i get it. well you know you asked this one at school and you drew the whole thing. only problem was i didnt think there would be any other double bond in the chain but only in the cyclohexene ring. But it was supposed to be the opposite. well thanks.Did it for you (sorry, diagram doesn't really look good)-

Then

P.S. diagram's not too bad!
- Messages
- 339
- Reaction score
- 105
- Points
- 53
Defend nhin kr rhihan han.. kar lo defend apnay aap ko...
mujhey pata tha k aap ko yehi lagey ga
