- Messages
- 112
- Reaction score
- 18
- Points
- 28
Guidance on how to solve these question.
17. 71.0 g of chlorine, Cl 2, react with an excess of sodium hydroxide solution at a particular temperature. The reaction produces exactly 35.5 g of product X.
What is product X?
A H 2O B NaCl C NaCl O D NaCl O3
8. Sulfur can be oxidised in two ways.
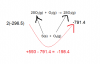
S(s) + O2(g) → SO2(g) ∆H o = –296.5 kJ mol–1 2S(s) + 3O2(g) → 2SO3(g) ∆H o = –791.4 kJ mol–1
Sulfur trioxide can be made from sulfur dioxide and oxygen.
2SO2(g) + O2(g) → 2SO3(g)
What is the standard enthalpy change for this reaction?
A –1384.4 kJ mol–1 B –989.8 kJ mol–1 C –494.9 kJ mol–1 D –198.4 kJ mol–1
17. 71.0 g of chlorine, Cl 2, react with an excess of sodium hydroxide solution at a particular temperature. The reaction produces exactly 35.5 g of product X.
What is product X?
A H 2O B NaCl C NaCl O D NaCl O3
8. Sulfur can be oxidised in two ways.
S(s) + O2(g) → SO2(g) ∆H o = –296.5 kJ mol–1 2S(s) + 3O2(g) → 2SO3(g) ∆H o = –791.4 kJ mol–1
Sulfur trioxide can be made from sulfur dioxide and oxygen.
2SO2(g) + O2(g) → 2SO3(g)
What is the standard enthalpy change for this reaction?
A –1384.4 kJ mol–1 B –989.8 kJ mol–1 C –494.9 kJ mol–1 D –198.4 kJ mol–1